Assignment Help With Wettability
Wettability
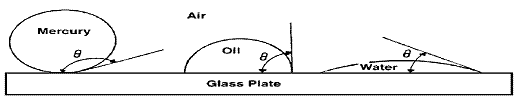
Wettability is defined as the tendency of one fluid to spread on or adhere to a solid surface in the presence of other immiscible fluids. The tendency of a liquid to spread over the surface of a solid is an indication of the wetting characteristics of the liquid for the solid. This spreading tendency can be expressed more conveniently by measuring the angle of contact at the liquid-solid surface. This angle, which is always measured through the liquid to the solid, is called the contact angle q. The contact angle q has achieved significance as a measure of wettability. Complete wettability would be evidenced by a zero contact angle, and complete non wetting would be evidenced by a contact angle of 180°. There have been various definitions of intermediate wettability but, in much of the published literature, contact angles of 60° to 90° will tend to repel the liquid. The wettability of reservoir rocks to the fluids is important in that the distribution of the fluids in the porous media is a function of wettability. Because of the attractive forces, the wetting phase tends to occupy the smaller pores of the rock and the non wetting phase occupies the more open channel.
Surface And Interfacial Tension
In dealing with multiphase systems, it is necessary to consider the effect of the forces at the interface when two immiscible fluids are in contact. When these two fluids are liquid and gas, the term surface tension is used to describe the forces acting on the interface. When the interface is between two liquids, the acting forces are called interfacial tension.
Surfaces of liquids are usually blanketed with what acts as a thin film. Although this apparent film possesses little strength, it nevertheless acts like a thin membrane and resists being broken. This is believed to be caused by attraction between molecules within a given system. All molecules are attracted one to the other in proportion to the product of their masses and inversely as the squares of the distance between them.
Email Based Assignment Help in General Composition Of Petroleum
To submit General Composition Of Petroleum assignmentClick here.
Following are some of the topics in General Composition Of Petroleum in which we provide help:
- General Composition Of Petroleum
- Physical Properties Of Hydrocarbons
- Origin of Petroleum
- Fundamental properties Of Fluid Permeated Rocks
- Porosity
- Permeability
- The Klinkenberg Effect
- Saturation
- Wettability
- Capillary Pressure
- Relative Permeability
- Drainage Process
- Three phase Relative Permeability
- Rock Compressibility
- Fundamentals Of Reservoir Fluid Behavior
- Classification Of Reservoir And Reservoir Fluids
- Gas Reservoirs
- Fundamentals Of Reservoir Fluid Flow
- Types Of Fluids
- Properties Of natural Gases
- Behavior Of Ideal Gases
- Behavior of Real Gases
- Compressibility Of Natural Gases
- Properties Of Crude Oil Systems
- Gas Solubility
- Determination And Application of Reservoir Fluid Properties
- Composition Of The Reservoir Fluid
- Differential Liberation Test
- Separator Tests
- Fluid Analysis Data On Gas
- Constant-Volume Depletion
- Oil Recovery mechanisms And The material Balance Equation
- Primary Recovery Mechanisms
- The Depletion Drive Mechanism
- Gas Cap Drive
- The Water Drive Mechanism
- Water Production
- The Gravity-Drainage-Drive Mechanism
- The Combination-Drive Mechanism
- The Material Balance Equation
- Change in Pore Volume Due to Initial Water and Rock Expansion
- Gas Reservoirs Help
- The Volumetric Method
- The material Balance Method
Petroleum Engineering | Petroleum Engineering Courses | Rotary Drilling | Gas Reservoirs | Behavior Of Ideal Gases | Online Tutoring