Composition Of The Reservoir Fluid Assignment Help
Composition Of The Reservoir Fluid
It is desirable to obtain a fluid sample as early in the life of a field as possible so that the sample will closely approximate the original reservoir fluid. Collection of a fluid sample early in the life of a field reducesthe chances of free gas existing in the oil zone of the reservoir.Most of the parameters measured in a reservoir fluid study can be calculated  with some degree of accuracy from the composition. It is the most complete description of reservoir fluid that can be made. In the past, reservoir fluid compositions were usually measured to include separation of the component methane through hexane, with the heptanes and heavier components grouped as a single component reported with the average molecular weight and density. With the development of more sophisticated equations-of-state to calculate fluid properties, it was learned that a more complete description of the heavy components was necessary. It is recommended that compositional analyses of the reservoir fluid should include a separation of components through C10 as a minimum.
with some degree of accuracy from the composition. It is the most complete description of reservoir fluid that can be made. In the past, reservoir fluid compositions were usually measured to include separation of the component methane through hexane, with the heptanes and heavier components grouped as a single component reported with the average molecular weight and density. With the development of more sophisticated equations-of-state to calculate fluid properties, it was learned that a more complete description of the heavy components was necessary. It is recommended that compositional analyses of the reservoir fluid should include a separation of components through C10 as a minimum.
Constant Composition Expansion Tests(Flas Liberation Test)
Constant-composition expansion experiments are performed on gas condensates or crude oil to simulate the pressure-volume relations of these hydrocarbon systems. The test is conducted for the purposes of determining:
- Saturation pressure (bubble-point or dew-point pressure)
- Isothermal compressibility coefficients of the single-phase fluid in excess of saturation pressure
- Compressibility factors of the gas phase
- Total hydrocarbon volume as a function of pressure
Composition Of The Reservoir Fluid Assignment Help By Online Tutoring and Guided Sessions from AssignmentHelp.Net
![]()
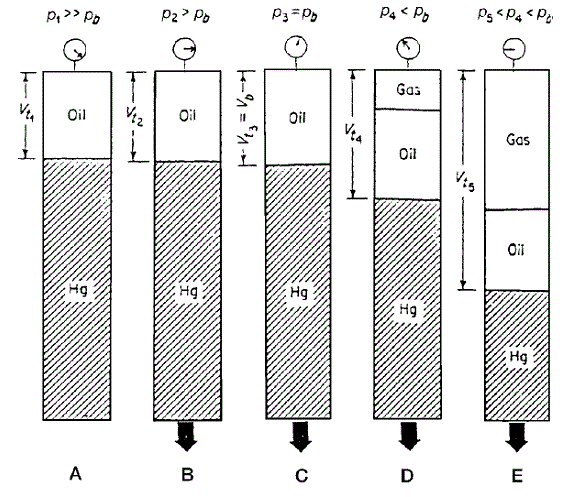
The experimental procedure, as shown schematically above involves placing a hydrocarbon fluid sample (oil or gas) in a visual PVT cell at reservoir temperature and at a pressure in excess of the initial reservoir pressure (Figure, Section A). The pressure is reduced in steps at constant temperature by removing mercury from the cell, and the change in the total hydrocarbon volume Vt is measured for each pressure increment.
The saturation pressure (bubble-point or dew-point pressure) and the corresponding volume are observed and recorded and used as a reference volume Vsat (Figure, Section C). The volume of the hydrocarbon system as a function of the cell pressure is reported as the ratio of the reference volume. This volume is termed the relative volume and is expressed mathematically by the following equation
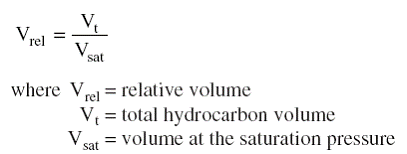
The relative volume is equal to one at the saturation pressure. This test is commonly called pressure-volume relations, flash liberation, flash vaporization, or flash expansion.
It should be noted that no hydrocarbon material is removed from the cell, thus, the composition of the total hydrocarbon mixture in the cell remains fixed at the original composition.
Email Based Assignment Help in Composition Of The Reservoir Fluid
To submit Composition Of The Reservoir Fluid assignment click here.
Following are some of the topics in General Composition Of Petroleum in which we provide help:
- General Composition Of Petroleum
- Physical Properties Of Hydrocarbons
- Origin of Petroleum
- Fundamental properties Of Fluid Permeated Rocks
- Porosity
- Permeability
- The Klinkenberg Effect
- Saturation
- Wettability
- Capillary Pressure
- Relative Permeability
- Drainage Process
- Three phase Relative Permeability
- Rock Compressibility
- Fundamentals Of Reservoir Fluid Behavior
- Classification Of Reservoir And Reservoir Fluids
- Gas Reservoirs
- Fundamentals Of Reservoir Fluid Flow
- Types Of Fluids
- Properties Of natural Gases
- Behavior Of Ideal Gases
- Behavior of Real Gases
- Compressibility Of Natural Gases
- Properties Of Crude Oil Systems
- Gas Solubility
- Determination And Application of Reservoir Fluid Properties
- Composition Of The Reservoir Fluid
- Differential Liberation Test
- Separator Tests
- Fluid Analysis Data On Gas
- Constant-Volume Depletion
- Oil Recovery mechanisms And The material Balance Equation
- Primary Recovery Mechanisms
- The Depletion Drive Mechanism
- Gas Cap Drive
- The Water Drive Mechanism
- Water Production
- The Gravity-Drainage-Drive Mechanism
- The Combination-Drive Mechanism
- The Material Balance Equation
- Change in Pore Volume Due to Initial Water and Rock Expansion
- Gas Reservoirs Help
- The Volumetric Method
- The material Balance Method


