Enzymology Assignment Help
Enzymology And Enzyme Technology
Enzymes are basically biological catalysts, which is central to every biochemical process, acting in organized sequences, they catalyze the hundreds of stepwise reactions that degrade nutrients molecules, conserve and transform chemical energy, and make biological molecules from simple macromolecules.
With the exception of some of them such as catalytic RNAs, all we knows that enzymes are proteins. Many require nonprotein co-enzymes or cofactors for their catalytic function which are known as prosthetic groups. A complete catalytically active enzyme together with its bound coenzyme and/or metal ions is called a holoenzyme. The protein part of such an enzyme is called the apoenzyme or apoprotein.
Enzymes are classified according to the type of reactions they catalyze. All enzymes have formal E.C numbers and names, and most have trivial names. The classes of enzymes are oxidoreductases, transferases, hydrolases, lyases, isomerases, and ligases.
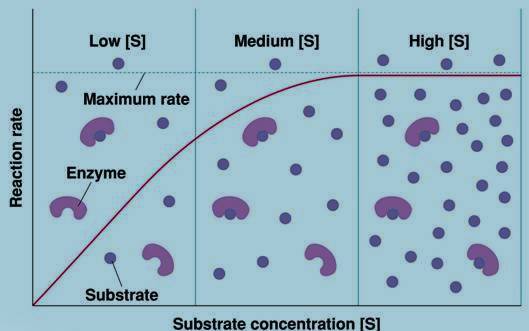
The distinguishing feature of an enzyme-catalyzed reaction is that it takes place within the confines of a pocket on the enzyme called the active site. The molecule that is bound to the active site and is acted upon by the enzyme is called the substrate.
Enzymes do not affect the reaction equilibria, their function is to increase the rate of reaction. Covalent interactions between the enzymes and the substrates lower the activation energy (and thereby accelerate the reaction) by providing an alternative, lower-energy reaction path.
Binding energy is a major source of free energy used by enzymes to lower the activation energies of reactions.
Enzymology Assignment Help By Online Tutoring and Guided Sessions from AssignmentHelp.Net

Email Based Homework Help in Enzymology And Enzyme Technology
To submit Enzymology And Enzyme Technology assignment click here


