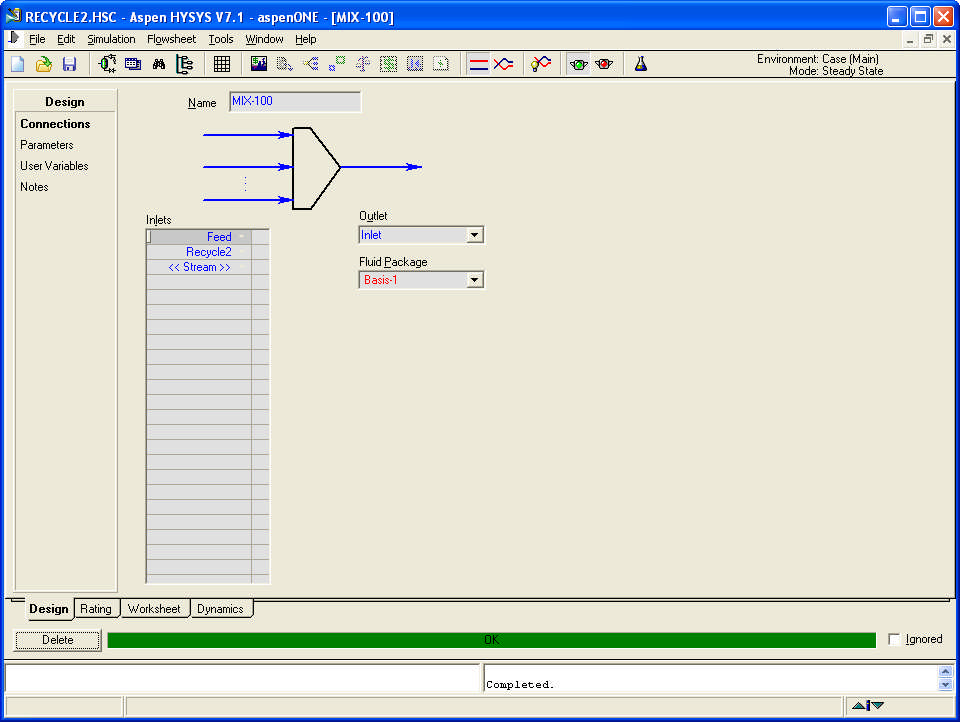
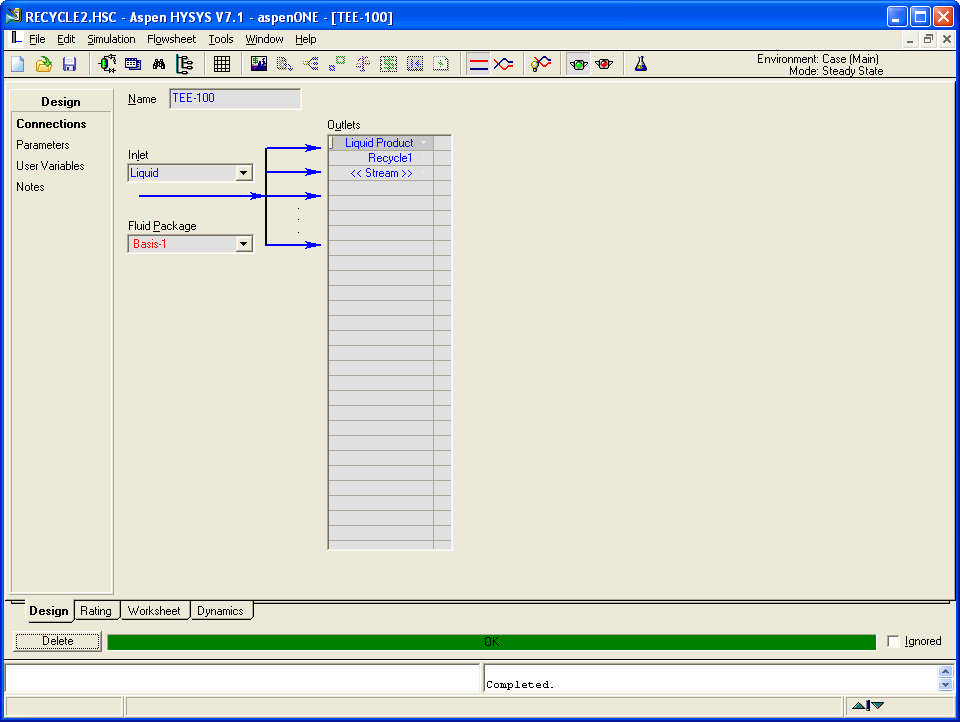
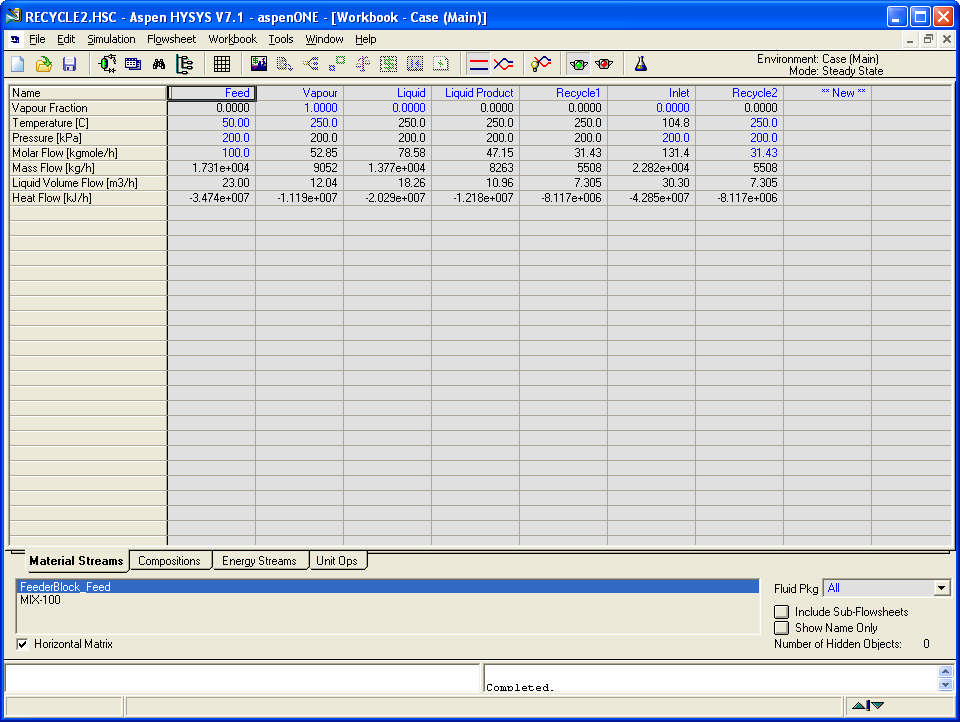
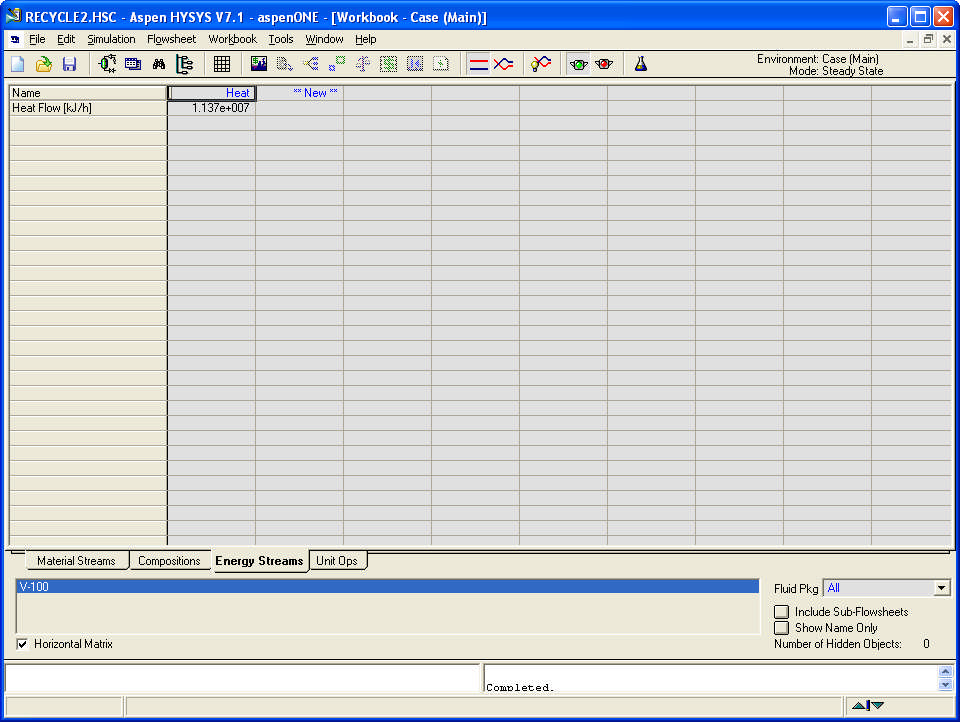
Compositions and energy streams

SCHOOL OF CHEMICAL AND PROCESS ENGINEERING
Dr. Antonia Borissova
2015/16
1. R.M. Felder & R.W. Rousseau,
Elementary Principles of
Chemical Processes, Third edition, John Wiley & Sons,
3. R.H. Perry, D.W. Green, Perry's Chemical Engineers Handbook, McGraw-Hill Education (ISE Editions), 1998.
4. Aspen HYSYS v7.3 (Help)
The General Balance Equation
input + generation – output – consumption = accumulation input (enters through system boundaries);
generation (produced within system);
output (leaves through system boundaries);
consumption (consumed within system);
accumulation (buildup within system.Balance on a conserved quantity:
Non-reacting systems:
Units for material streams – mol (kmol); g (kg)
Remember:
input – output = accumulation
In batch processes you may have temporary accumulation, but not over the long termSome basic advice:
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| • n |
in | = | • n |
out | + | • n |
||||
| Batch: | n in | = | n out | + | ||||||
0.316 mol T/mol |
|
|---|---|
Total mass balance:
| 5.0 | mol | = | .0 684n V | + | 4.0 n | ||||
|---|---|---|---|---|---|---|---|---|---|
| n V | |||||||||
| = | .0 352 mol ; n | L | = |
|
|||||
Enthalpy Table
Reference conditions: B(1, 10ºC, 1 atm); T(1,10ºC, 1 atm)
| 1 | = | 50°C ∫ |
(C | = |
|
6 | ( ) | dT | = | 37.53 kJ / mol | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
2 | 78 |
( ) |
|
= | |||||||||||||||||||||||||||||||||||||||
| = | 66
|
( ) | dT | + | ∆ | ∧ H |
v | | C | 6 | H | 6 | ( | 80 1.° | C | ) | + | p | )C H 6 | |||||||||||||||||||||||||
| 4 | = | 110 .62 C ∫(C |
( ) | dT | + | v | | C 7 | H | 8 | (110.62 C | ) | + | 50°C ∫(C |
p | )C H 7 | 8 | ( ) | dT | = | 42.93 kJ / mol | |||||||||||||||||||||||
5
| n-pentane |
|
|
||
|---|---|---|---|---|
|
|
|||
|
calculate:
(e) mole rates and enthalpies of all streams;
(f) the required rate of heat addition to the evaporator.
Raoult' s law for pentane and hexane
| 0.410 | p | * P |
( | 65 | o | C) = | yP 0 | y | = | 0 656 |
|
||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.590 | p | * H |
( | 65 | o | C) = (1 | − | ⇒ | P 0 | = | 1157 | ||||
Ideal Gas Equation
|
V | = | n RT v | = | = | 667 | |||
|---|---|---|---|---|---|---|---|---|---|
| v | P 0 |
| ∧ H |
( ) | = | T b ∫ |
C dT pl | + | + | ∫T b | C dT pv | ∧ H |
( ) | = |
|
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
∧ H |
( ) | = | ||||||||||||||||||||||||||
| i.e. | ∧ H |
( ) | = | ∫ 65 |
C dT pl | + | + | C dT pv | and | ||||||||||||||||||||
Energy Balance Equation
|
= | ∑ | • n |
out | ∧ H out |
− | ∑ | = |
|---|
Systems with recycles
Task 1 (cont.)
If the product is the vapour stream, the liquid stream is recycled.
|
|||
|
|||
|
|||
Recycles are used for
Recovery and reusing unconsumed reactants Recovery of catalyst
Dilution of a process stream
Control of a process variable
Circulation of a working fluid
9
Task 2: Mass & Energy Balances of a System with One Evaporator
Problem:
Part of the liquid outlet from the separator (40%) is recycled.
10
12
14
16
18
2,3-dimethyl pent-2-ene 0.3
2,3,3,-trimethyl pent-1-ene 0.4
Write a technical report (up to 1500 words excluding graphical material) using MS Word to describe your approach to the solution of the task, including the problem statement, the structural formulae of the three components, reason for the selection of the Equation of State, problem background and methodology applied. Show the HYSYS PFD and the Workbook screens: Material Streams, Compositions and Energy Streams. Analyse the PFD for the process conditions as set in the assignment. Comment on the advantages and disadvantages of the two systems –with one and with two separators. Study the sensitivity of the one separator process to changes in temperature only and of the two separator process to changes in temperature, pressure and recycle ratio.
Submit the report electronically to the VLE.