Proteomics Assignment Help
What Is Proteomics?
- Proteomics is the study of the proteome—the "PROTEin complement of the genOME"
- More specifically, "the qualitative and quantitative comparison of proteomics under different conditions to further unravel biological processes"
What Makes Proteomics Important{'?'}

- A cell's DNA - its genome - describes a blueprint for the cell's potential, all the possible forms that it could conceivably take. It does not describe the cell's actual, current form, in the same way that the source code of a computer program does not tell us what input a particular user is currently giving his copy of that program.
- All cells in an organism contain the same DNA.
- This DNA encodes every possible cell type in that organism-muscle, bone, nerve, skin, etc.
- If we want to know about the type and state of a particular cell, the DNA does not help us, in the same way that knowing what language a computer program was written in tells us nothing about what the program does.
- There are more than 160,000 genes in each cell, only a handful of which actually determine that cell's structure.
- Many of the interesting things about a given cell's current state can be deduced from the type and structure of the proteins it expresses.
- Changes in, for example, tissue types, carbon sources, temperature and stage in life of the cell can be observed in its proteins.
Proteomics in Disease Treatment
- Nearly all major diseases—more than 98% of all hospital admissions—are caused by a particular pattern in a group of genes.
- Isolating this group by comparing the hundreds of thousands of genes in each of many genomes would be very impractical.
- Looking at the proteomes of the cells associated with the disease is much more efficient. Many human diseases are caused by a normal protein being modified improperly. This also can only be detected in the proteome, not the genome.
- The targets of almost all medical drugs are proteins. By identifying these proteins, proteomics aids the progress of pharmacogenetics.
Proteomics Assignment Help By Online Tutoring and Guided Sessions from AssignmentHelp.Net
Examples
What do these have in common?
- Alzheimer's disease
- Cystic fibrosis
- Mad Cow disease
- An inherited form of emphysema
Protein Folding
What is it?
- Fundamental components
- Proteins
- Ribosome's string together long linear chains of amino acids.
- Called Proteins
- Loop about each other in a variety of ways
- Known as folding
- Determines whether or not the protein functions

Dangers
- Folding determines function
- Of the many ways of folding one means correct functionality
- Mis-folded proteins can mean the protein will have a lack of functionality
- Even worse can be damaging or dangerous to other proteins
- Too much of a mis-folded protein can be worse than too little of a normal folded one
- Can poison the cells around it
- Mis-folded proteins can mean the protein will have a lack of functionality
History
- Linus Pauling — half a century ago
- Discovered
- A-helix
- B-sheets
- These are found in almost every protein
- Discovered
- Christian Anfinsen — early 1960's
- Discovered
- Proteins tie themselves
- If separated fold back into their own proper form
- No folder or shaper needed
- Discovered
Expansion to Anfinsen
- Sometime the protein will fold into the WRONG shape
- Chaperones
- Proteins who's job is to keep their target proteins from getting off the right folding path
- These two key elements help us understand keys to protein folding diseases
What is Protein Folding?
- Primary Structure
- 3-D conformation of a protein depends only on its linear amino acid sequence
- In theory can be computed explicitly with only this information
- One of the driving forces that is thought to cause protein folding is called the hydrophobic effect
Hydrophobic effect
- Certain side chains do not like to be exposed to water
- Tend to be found at the core of most proteins
- Minimize surface area in contact with water
Proteins
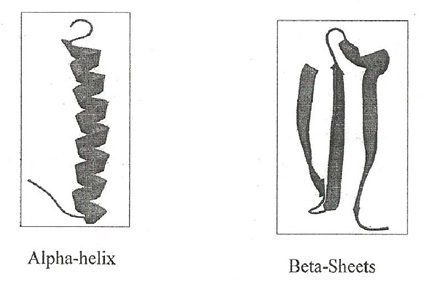
- Two Repetitive features of a protein
- Alpha-helix
- Beta-sheet
- Alpha-helix
- consecutive residues
- Arranged in spiral staircase
- Beta-Sheets
- Comprised of two or more extended strands of amino-acids joined by inter strand hydrogen bonds
Hydrogea Bonds
- In both secondary structures
- Alpha-helix
- Beta-Sheets
- Responsible for stabilization
- Greatly affect the final fold of the protein
Fold Calculation
- Of all the possible ways the protein could fold, which one is
- Most stable structure
- Lowest energy
- Calculation of protein energy is only approximate
- Thus compounding the complexity of such a calculation
- Requiring enormous computational power
Why Fold Proteins
- Mans genetic diseases are caused by dysfunctional proteins
- Build better cures
- Understand mutation
- Assign structures functions to every protein
- Thus understand the human genome
- Decode the Human DNA
