Chemistry Assignment Help With Pyrolysis Of Esters

Pyrolysis Of Esters
Thermal cleavage of an ester usually involves the formations of a six membered ring as the transition state leading to the elimination of acid leaving behind alkene.
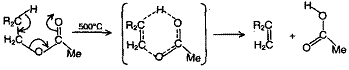
As a direct consequence of cyclic transition state, both the leaving groups namely proton and carboxylate ion are in the cis position. This is an example of cis elimination.
Homework Help For Pyrolysis Of Esters
assignmenthelp.net provides best Online Assignment Help service in chemistry for all standards. Our Tutor provide their high quality and optimized Tutorial help to fulfill all kind of need of Students.
To submit Pyrolysis Of Esters assignment click here.
