Monomers Assignment Help
Monomers
It is substances, which have capacity to forming covalent bonds with a sequences of additional like or unlike molecules within condition of relevant polymers forming reaction for particular case. Or we can say that these are the substances which play important role in polymerization reactions. These are single molecules and due this called monomers. It can be monosaccharide, which gives after reaction polysaccharide. Amino acids are mono protein molecule which gives polypeptide after reactions, and these polypeptide become protein after folding.
Amino acids are natural monomers, which are basically found at nucleus, and then form nucleic acids DNA, RNA by the process of polymerization. Starch, glycogen and cellulose etc are gives glucose which is also natural monomers. Xylose is a byproduct of polymerization reaction of xylan (natural monomers), here another example of natural monomer called isoprene and after polymerization it gives rubber.
Application: Propylene is used in membrane based recovery process. Due to polyfunctionality, which is essential features of monomer, used in making polymers.
Monomers Assignment Help By Online Tutoring and Guided Sessions at AssignmentHelp.Net
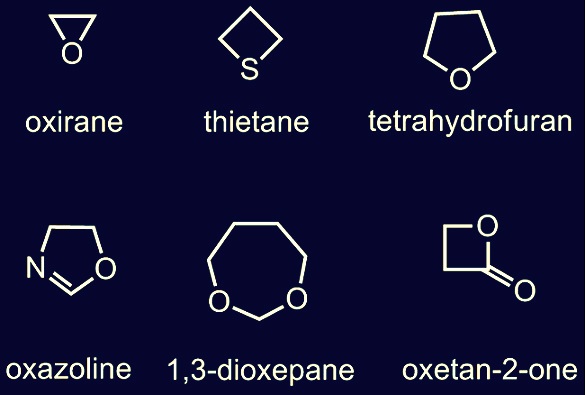
Addition reactions are basic characteristic of monomers, which contain either a double bond between two molecule or a ring like structures which may be heteromorphic or usually three to seven atoms. For monomers condensation reaction is typical, in which two or more atomic groups are available, such as compound which is both alcohol and acid can go repetitive ester formations through involving alcohol group of each molecule to the every acid group molecules, and it makes chain of molecules called polyester.

Email Based Assignment Help in Monomers
To submit Monomers assignment Click here.
