Chemistry Homework Help With Preparation Of Alkanes

Methods Of Preparation Of Alkanes
This may be done in three different ways
Reduction by dissolving metals
Reduction by dissolving metals e.g. zinc and acetic or hydro chloric acid, zinc and sodium hydroxide, zinc-copper couple and ethanol, etc. In this reaction, earlier ‘nascent’ hydrogen was considered to be the reducing agent.
Zn ® Zn2+ + 2e–
RX + e– ® X– + R· R:–
R:– + C2H5OH ® R – H + –OC2H5
Reduction by reducing agents like LIAIH4, NaBH4 etc.:
Primary and secondary alkyl halides are readily reduced to alkanes bv lithium aluminium hydride (LiAIH4) while reduction of tertiary halides with LiAIH4 gives mainly alkenes. On the other hand, sodium borohydrides (NaBH4) reduces secondary and tertiary halides. but not primary, whereas triphenylin hydride (Ph3SnH) reduces all three types of alkyl halides. So each reducing agent is specific in its action.
(a) 4R – X + LiAlH4 ® 4R-H + LiX + AlX3(X ¹F)
or R-X + H:(–) ® R-H + X– (H comes from LiAlH4)
(b) R-X + (n – C4H9)3SnH ®R-H + (n - C4H9)3SnX
Using organometallic compounds like Grlgnard Reagent:
Alkyl halides react with either Mg or Li in dry ether to give organometallic compounds having a basic carbanionic site.
![]() then
then ![]()
![]() then
then ![]()
The net effect is replacement of X by H. This reaction can be afforded with any compound that is more acidic than alkane e.g., alcohols, NH3, terminal alkynes etc. Thus the net effect of the reaction is the displacement of a weak acid from its salt by a strong acid. Using this reaction, we determine the number of active hydrogens present in a given compound (the one which reacts with Grignard reagent).
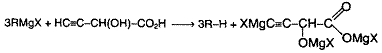
Assignment Help For Preparation Of Alkanes
assignmenthelp.net provides best Online Assignment Help service in Alkanes for all standards. Our Tutor provide their high quality and optimized Tutorial help to fulfill all kind of need of Students.
To submit Preparation Of Alkanes assignment click here.
Hydrocarbons Homework Help | Assignment Help | Homework For Chemistry | homework help | Homework Assignment Help | Online tutoring services | Term paper help | Online Tutoring


