Enols, Enolate anions, Enamines Assignment Help
Introduction to Enols, Enolate anions, Enamines:
Enols are vinyl alcohols, C=C-OH. It is less stable than the keto form. The keto-enol tautomerization is catalyzed by acids or bases. This makes enols viable intermediates in many important reactions.
The most important reaction of enolate anions is nucleophilic addition to the carbonyl group of another molecule of the same or different compound although these reactions may be catalyzed by either acid or base, base catalysis is more common. Enolates are carbanions at the carbon to a carbonyl group; Enolates are powerful nucleophiles with the negative charge spread over a carbon and the oxygen of the carbonyl.
Enamines are vinyl amines, C=C-N.

Enols, Enolate anions, Enamines Assignment Help By Online Tutoring and Guided Sessions at AssignmentHelp.Net
We are presenting a short note on the Enols, Enolate anions, Enamines for references purpose for all students. Assignment Help, project help, homework help, and online help are also available. Online chatting and online tutorial with our expert team are the key feature help of assignmenthelp.net.
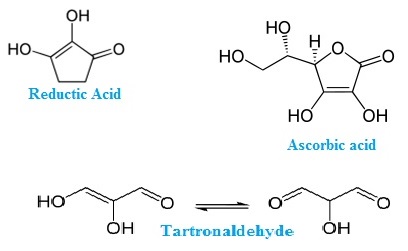
Examples of reductones for Enols:
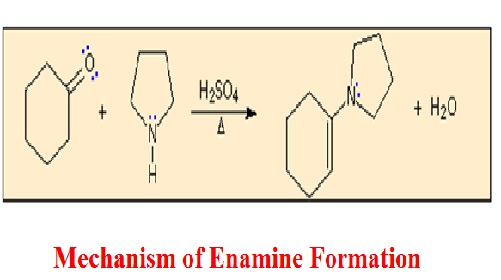
The Mechanism of Enamine Formation:
Formation of enolate anion:
- Formed by treating aldehyde, ketone, or ester (each which has at least 1 alpha-H)
w/ base - -most of negative charge in enolate anion is on O
What kind of Assignment Help for Enols, Enolate anions, Enamines, We provide:
Small introduction to Enols, Enolate anions, Enamines is offered with proper explanation and screen shot of example. Assignmenthelp.net provides all possible help for the assignment work with Enols, Enolate anions, Enamines.
We have online tutorial facility for student who wishes to learn Enols, Enolate anions, Enamines. Anyone can join this service and learn the Enols, Enolate anions, Enamines. If any student needs help with the Enols, Enolate anions, Enamines, then you can chat with our expert tutor at a nominal cost. Our service is open to all and our goal is to provide help for everyone.
Email Based Assignment Help in Alkyl Halides, Ethers, Thiols and Sulfides
To submit Enols, Enolate anions, Enamines assignment click here


