Chemistry Assignment Help With Chemical Properties Of The Alkanes

Chemical Properties Of The Alkanes
The alkanes are generally stable towards common reagents at room temperature, the minimum energy required to cause homolytic cleavage of almost non-polar C-H and C-C bonds is not available at room temperature. However, alkanes undergo substitution reactions at high temperature. As the temperature is increased, the
C-H and C-C bonds break forming free radicals as intermediates. The reactivity of alkanes is decided on the basis of stability of the free radicals. As the stability of free radical increases, energy required producing them i.e. energy of activation decreases and hence the rate of reaction increases.
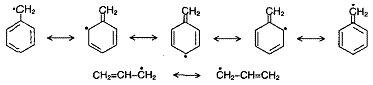
Benzyl radical (C6H5 -C·H2) and allyl radical (CH2=CH-C·H2) are stabilised by resonance.
We are offering the following main topics for assignment like:
Assignment Help For Chemical Properties of the Alkanes
assignmenthelp.net provides best Online Assignment Help service in Chemistry for all standards. Our Tutor provide their high quality and optimized Tutorial help to fulfill all kind of need of Students.
To submit Chemical Properties of the Alkanes assignment click here.
